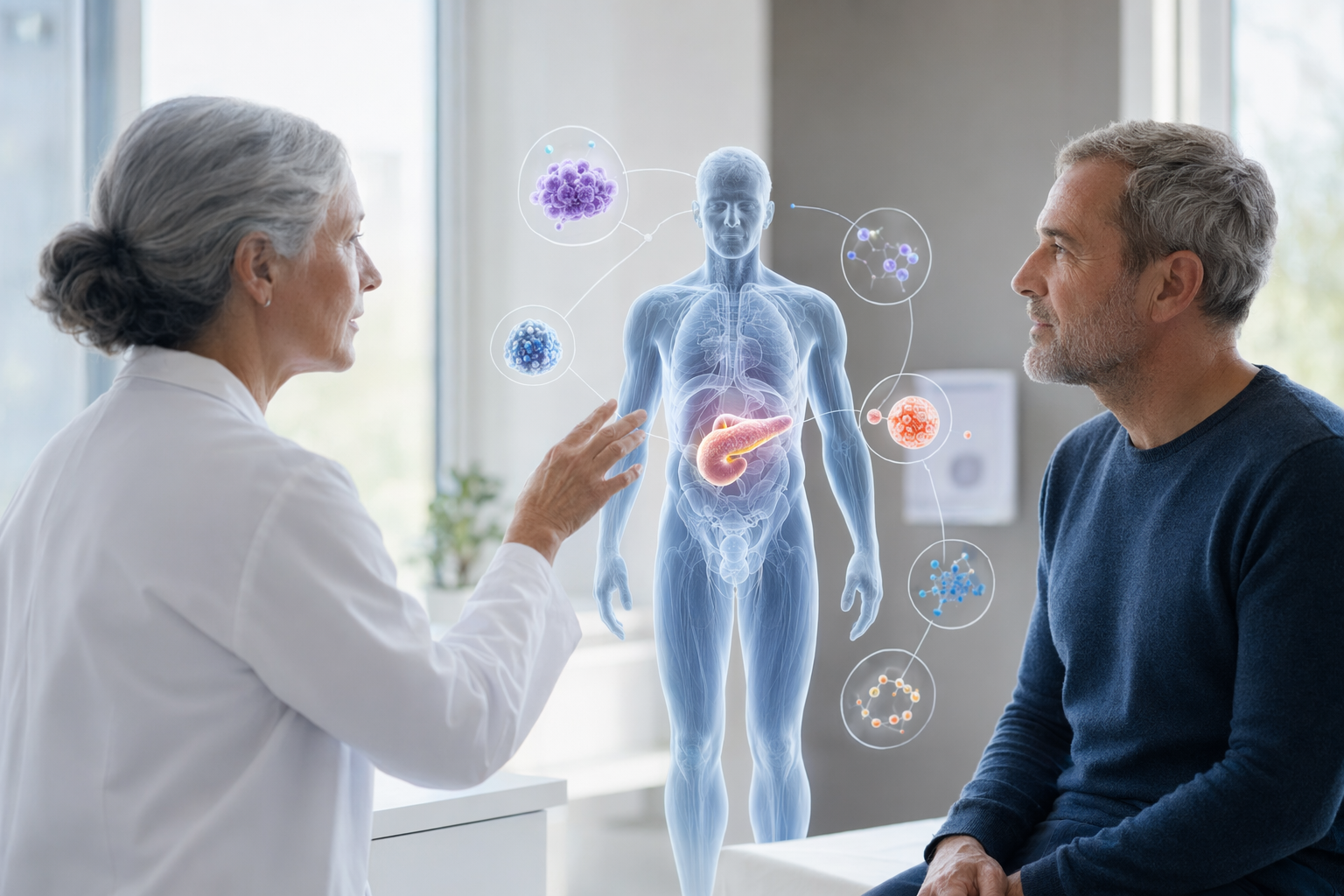
What it is
BioTwin's Cancer Screening Decision Assistance Tool (CDSS) is a screening decision assistance tool. It is not a screening tool. It is not a diagnostic device. It supports earlier and better-targeted screening conversations between you and your health professional, by giving them novel longitudinal information about your biology.

How it works
BioTwin combines biological markers (from at-home kits), wearable signals, lifestyle data, and history into a structured cancer risk profile across six cancers. The profile is shared with your health professional, who decides, based on their clinical judgment, whether further screening is appropriate.
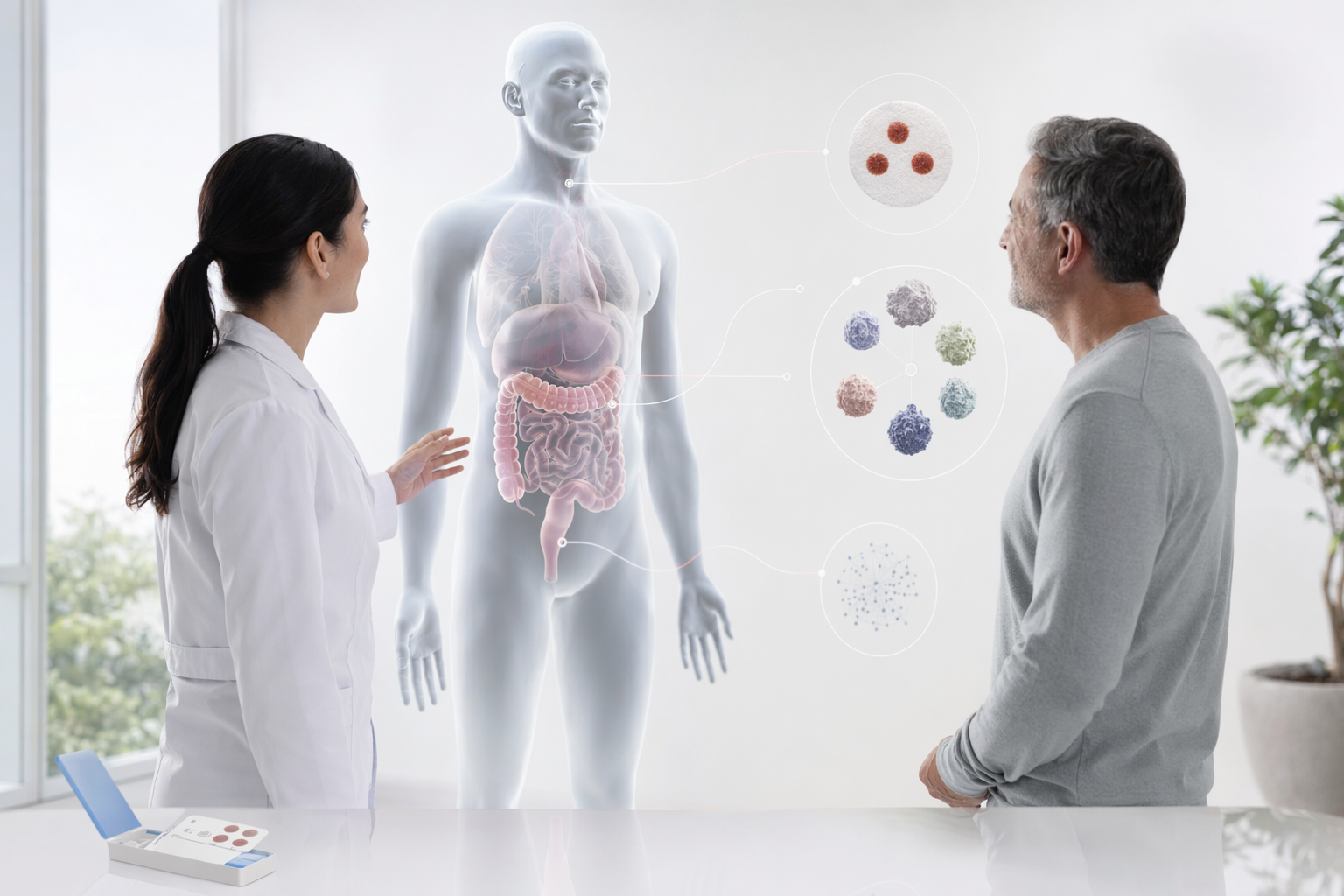
How BioTwin works with your health professional
BioTwin is not, and never will be, an expert in oncology. BioTwin's role is to provide novel longitudinal information that your health professional does not otherwise have access to. The clinical decision always remains with your health professional.
Where it is approved
Currently regulatory approved in the UAE. Pending Health Canada and FDA review.

Important boundaries
The CDSS is a screening decision assistance tool. It is not a screening tool. It is not a diagnostic device. It does not replace clinical judgment, screening, or diagnosis. Treatment and diagnosis decisions remain with licensed healthcare professionals.

For specific questions, contact BioTwin.